Separation and qualitative determination of cations and anions play a pivotal role in various scientific disciplines, enabling the identification and quantification of these charged species in complex samples. This article delves into the principles, techniques, and applications of cation and anion separation, providing a comprehensive overview for researchers, students, and practitioners.
From analytical chemistry to environmental monitoring and industrial processes, the separation and qualitative determination of cations and anions offer invaluable insights into the composition and behavior of matter.
Introduction
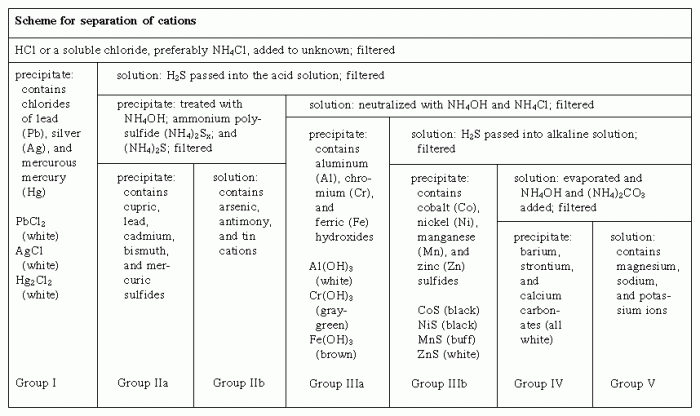
The separation and qualitative determination of cations and anions play a pivotal role in various fields, including analytical chemistry, environmental monitoring, and industrial processes. These techniques enable the identification and quantification of specific ions in complex samples, providing valuable information for a wide range of applications.
Principles of Separation Techniques
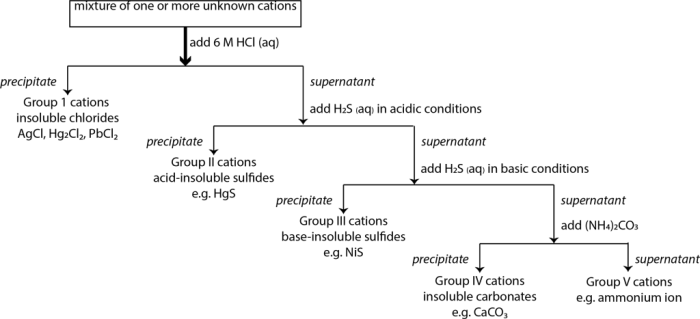
Precipitation
Precipitation is a widely used separation technique that involves the formation of an insoluble solid precipitate when a cation or anion reacts with a specific reagent. The precipitate can be filtered and analyzed to identify the corresponding ion.
Solvent Extraction, Separation and qualitative determination of cations and anions
Solvent extraction involves the selective transfer of ions from an aqueous solution to an immiscible organic solvent. The ions are extracted based on their solubility and complexation properties in the organic solvent.
Ion Exchange Chromatography
Ion exchange chromatography is a separation technique that utilizes ion exchange resins to separate ions based on their charge and affinity for the resin. The ions are adsorbed onto the resin and eluted with a suitable eluent.
Electrophoresis
Electrophoresis is a separation technique that utilizes an electric field to separate ions based on their charge and size. The ions migrate through a gel or solution under the influence of the electric field.
Qualitative Determination of Cations

Flame Tests
Flame tests involve heating a sample in a flame to produce a characteristic color. Different cations emit specific colors when excited in a flame, allowing for their identification.
Bead Tests
Bead tests involve mixing a sample with a flux and heating it on a loop of platinum wire. The color and appearance of the bead formed provide information about the presence of specific cations.
Spot Tests
Spot tests utilize reagents that produce specific colors or reactions when they come into contact with certain cations. These tests are performed on a filter paper or a spot plate.
Qualitative Determination of Anions: Separation And Qualitative Determination Of Cations And Anions
Precipitation Reactions
Precipitation reactions involve the formation of an insoluble precipitate when an anion reacts with a specific reagent. The precipitate can be identified based on its appearance and solubility properties.
Acid-Base Reactions
Acid-base reactions involve the reaction of an anion with an acid or a base. The resulting solution can be tested for pH changes or the formation of specific gases.
Specific Ion Tests
Specific ion tests utilize reagents that produce specific colors or reactions when they come into contact with certain anions. These tests are often used to detect specific anions in complex samples.
FAQ
What are the common methods for separating cations?
Common methods for separating cations include precipitation, solvent extraction, ion exchange chromatography, and electrophoresis.
How can anions be qualitatively determined?
Anions can be qualitatively determined using precipitation reactions, acid-base reactions, and specific ion tests.
What are the applications of cation and anion separation in environmental monitoring?
Cation and anion separation are used in environmental monitoring to assess water quality, soil contamination, and air pollution.